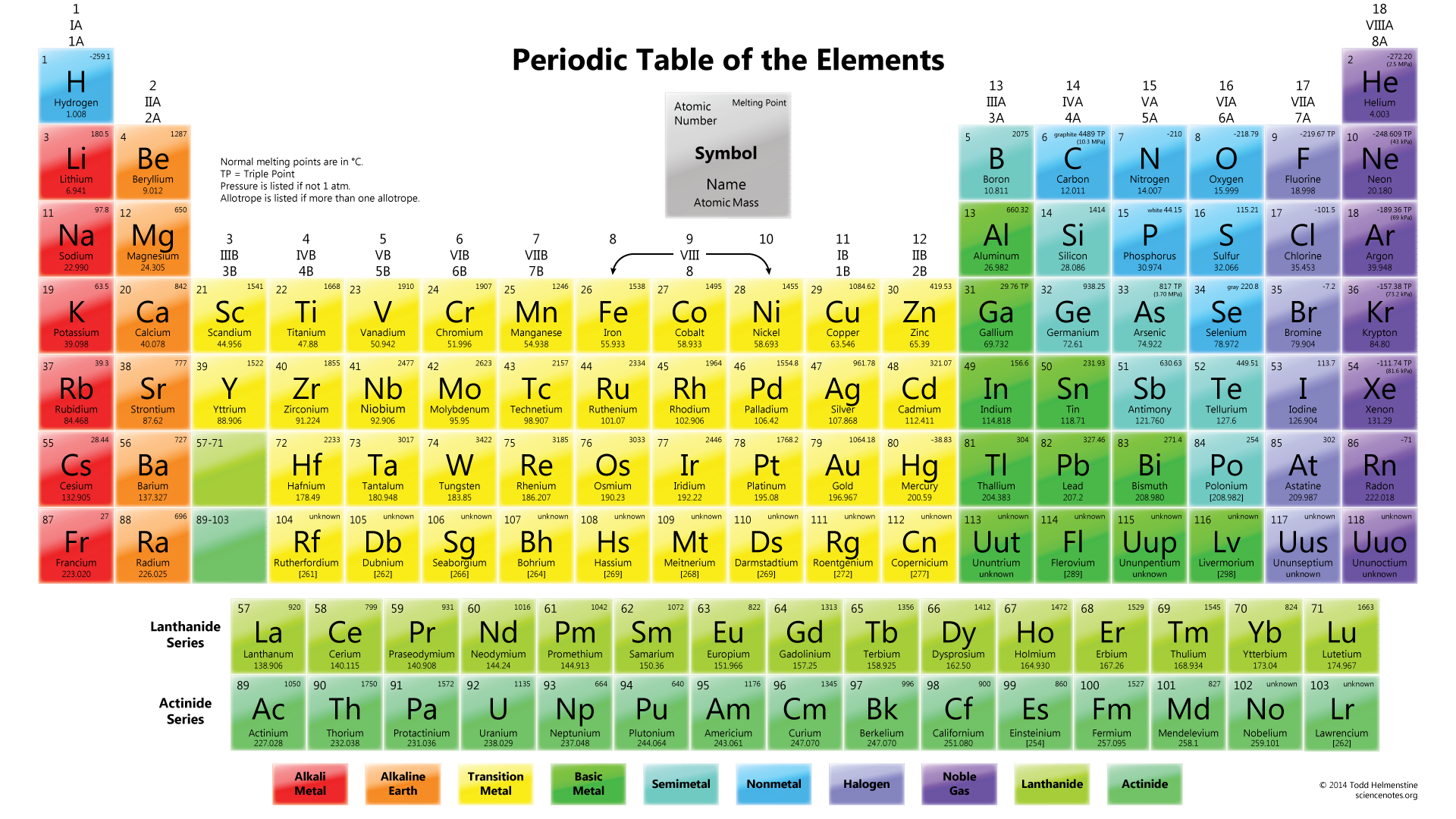 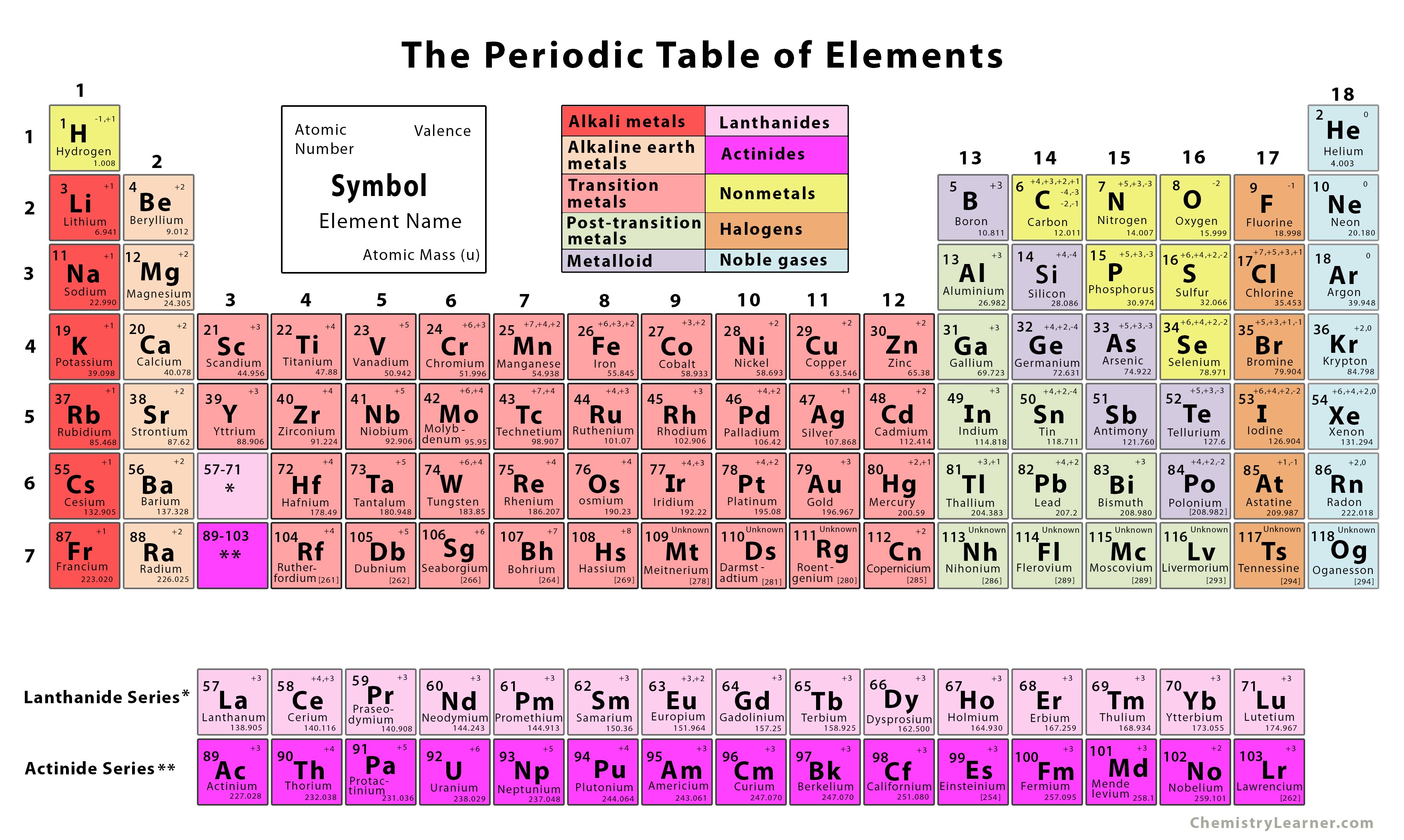
Bromine and iodine dissolve in organic solvents.Īs we move down the group the metallic nature of the elements increases. Iodine has a dark violet color.įluorine and chlorine are soluble in water. The elements of group 17 exhibit different colours. The physical properties of elements include:įluorine and chlorine are present in the gaseous state. In group 17, fluorine is the most electronegative element. The electronegativity decreases as we move down group 17 because of the increase in nuclear radii. The tendency of an atom to attract electron or a bonding pair of electrons is known as electronegativity. Attributable to the small size of the fluorine particle, the approaching electron encounters a more noteworthy measure of repulsion from the electrons that are now present. It is a result of the small size and reduced 2p sub-shell of the fluorine atom. among all, chlorine has the most extreme negative electron pick up enthalpy. chlorine has more negative electron pick up enthalpy than fluorine. This is attributed to the small size of fluorine due to which there is higher inter-electronic repulsion in the small 2p orbitals and lesser attraction for the incoming electron. However, on moving down the group the electron gain enthalpy turns out to be less negative.Įxception: chlorine has a higher gain of enthalpy when compared to fluorine. Halogens have negative electron gain enthalpy. The energy released when an electron is added to an isolated gaseous atom is known as electron gain enthalpy. The ionization energy of fluorine is comparatively higher than any other halogen which is due to its small size because of which greater will be the attraction between the core and the valence shell. This is because as we move down the group the size of the atom increases which decreases the attraction of the valence electrons and the valence electrons therefore only little ionization energy is required to remove an electron from the valence shell from any halogens. As we move down group 17, the ionization energy decreases. The energy required to remove an electron from its valence shell is known as ionization energy. The reason the elements belonging to this group have smaller atomic radii compared to other elements is due to high atomic charge. As we go down the group, the atomic and the ionic radii tend to increase as an extra energy shell is added. Ionic radii are the measure of the size of an atom's ion. The atomic properties of group 17 which are going to be discussed are:Ītomic radii are the measure of the distance from the center of an atom to the outermost shell containing electrons. Bromine exists as a liquid at room temperature. Chlorine, bromine, and iodine are present in the ocean water as chlorides, bromides, and iodides. Iodine exists as solid at room temperature. The dry bed of the ocean contains sodium chloride. Water in the ocean comprises 1.5% by weight of sodium chloride. It exists in its gaseous state at room temperature. Chlorine is the 20th richest element found by weight in the Earth's crust. Fluorine is also found in the soil, plants of stream water, and bones and teeth of creatures. It usually exists as insoluble fluorides, cryolites, fluorspar, and fluorapatite. It exists in the gaseous state at room temperature. Fluorine is the 13th richest element by weight in the crust of the world. Astatine is radioactive in nature and exists in a solid-state at room temperature. Halogens exist in all three different states of matter. Halogens do not exist in their free state. This is due to their tendency to pick up or share an electron to achieve an octet or closest inert gas configuration. Halogens are the most reactive non-metals. These elements require one more electron to achieve an octet or ideal gas configuration. The outermost shell is short by one electron to achieve an octet. There are 7 electrons in the outermost shell of the elements belonging to group 17. Therefore there are 7 electrons in the outermost shell. The electronic configuration of the outermost shell of the elements of group 17 is ns 2 and np 5. Group 17 elements, fluorine, chlorine, bromine, iodine, and astatine, are collectively known as halogens (in Greek, halo means salt, and genes mean generating, thus collectively salt-producing). 
These elements are grouped together as they have similar properties. Halogens are nonmetals with high reactivity. They are called halogens as they react with metals to produce salts. The elements that are present in group 17 are fluorine, chlorine, bromine, iodine, and astatine.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
